Manganese »
PDB 6i90-6jr1 »
6iht »
Manganese in PDB 6iht: Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide
Enzymatic activity of Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide
All present enzymatic activity of Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide:
3.1.3.16;
3.1.3.16;
Protein crystallography data
The structure of Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide, PDB code: 6iht
was solved by
C.-G.Yang,
T.Yang,
with X-Ray Crystallography technique. A brief refinement statistics is given in the table below:
| Resolution Low / High (Å) | 45.69 / 1.57 |
| Space group | P 1 21 1 |
| Cell size a, b, c (Å), α, β, γ (°) | 46.693, 38.609, 65.003, 90.00, 101.89, 90.00 |
| R / Rfree (%) | 15.9 / 18.3 |
Other elements in 6iht:
The structure of Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide also contains other interesting chemical elements:
| Magnesium | (Mg) | 2 atoms |
Manganese Binding Sites:
The binding sites of Manganese atom in the Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide
(pdb code 6iht). This binding sites where shown within
5.0 Angstroms radius around Manganese atom.
In total 4 binding sites of Manganese where determined in the Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide, PDB code: 6iht:
Jump to Manganese binding site number: 1; 2; 3; 4;
In total 4 binding sites of Manganese where determined in the Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide, PDB code: 6iht:
Jump to Manganese binding site number: 1; 2; 3; 4;
Manganese binding site 1 out of 4 in 6iht
Go back to
Manganese binding site 1 out
of 4 in the Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide
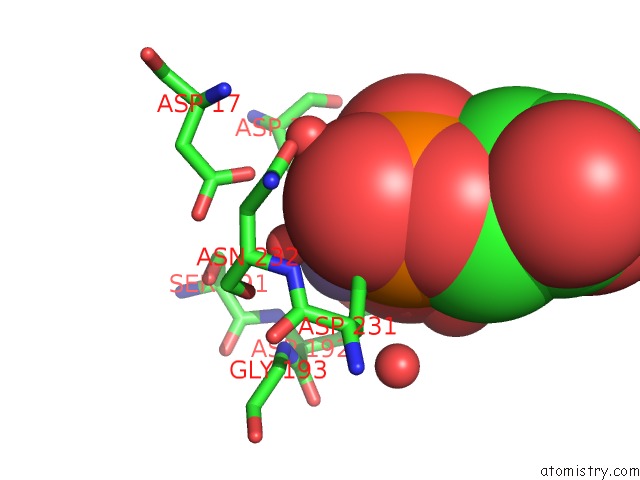
Mono view
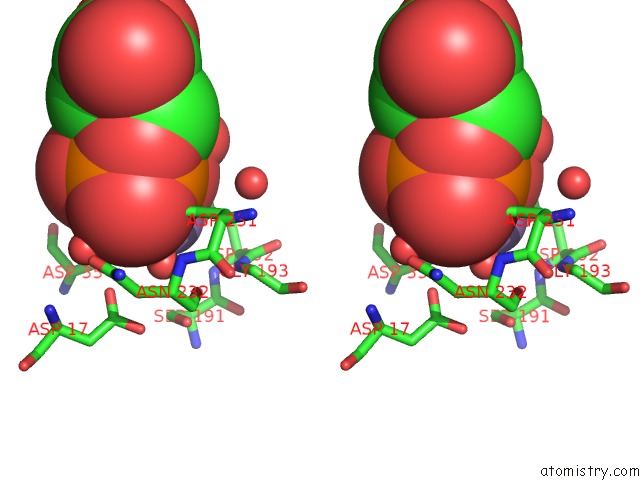
Stereo pair view

Mono view

Stereo pair view
A full contact list of Manganese with other atoms in the Mn binding
site number 1 of Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide within 5.0Å range:
|
Manganese binding site 2 out of 4 in 6iht
Go back to
Manganese binding site 2 out
of 4 in the Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide
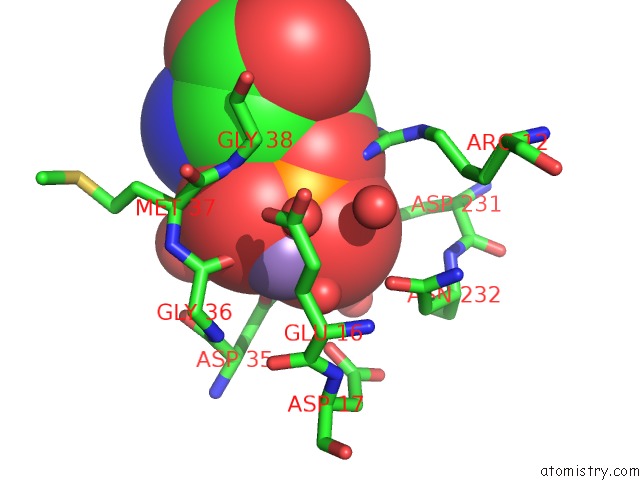
Mono view
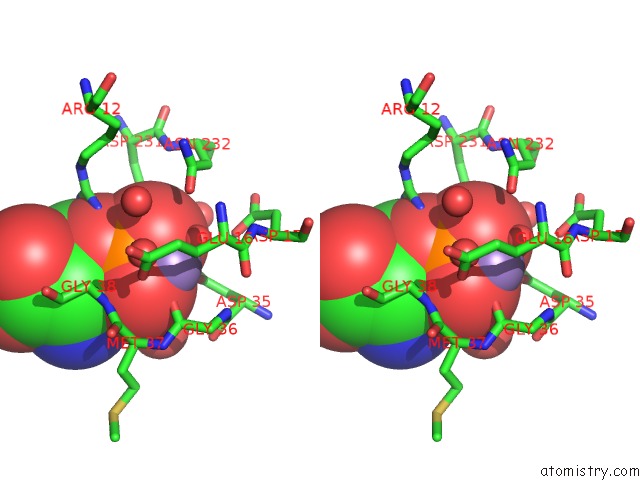
Stereo pair view

Mono view

Stereo pair view
A full contact list of Manganese with other atoms in the Mn binding
site number 2 of Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide within 5.0Å range:
|
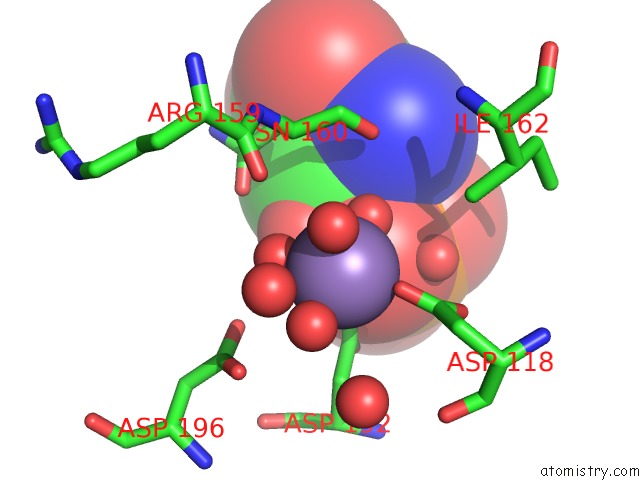
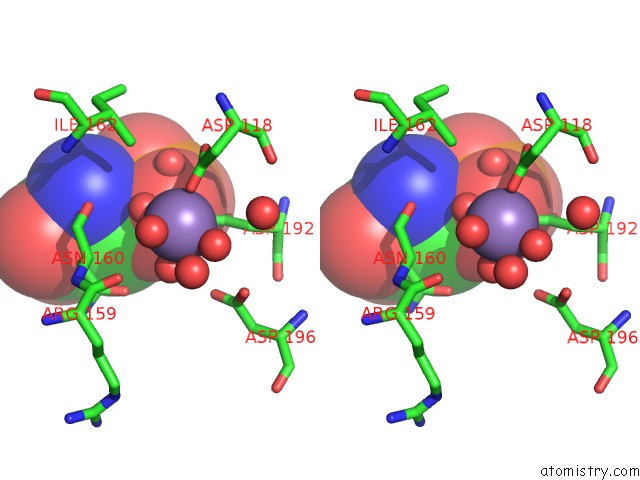
Manganese binding site 3 out of 4 in 6iht
Go back to
Manganese binding site 3 out
of 4 in the Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Manganese with other atoms in the Mn binding
site number 3 of Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide within 5.0Å range:
|
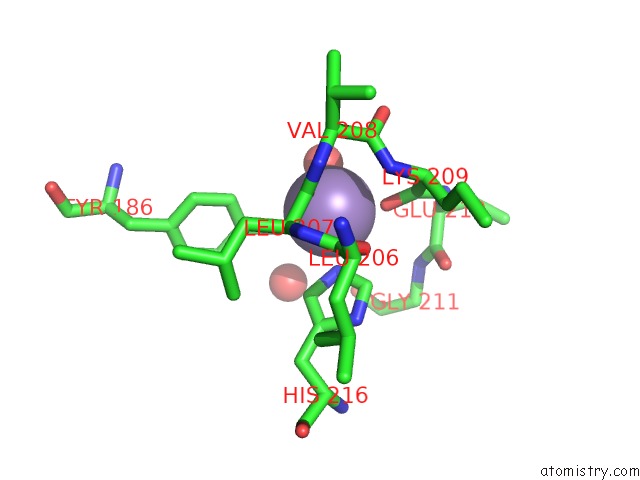
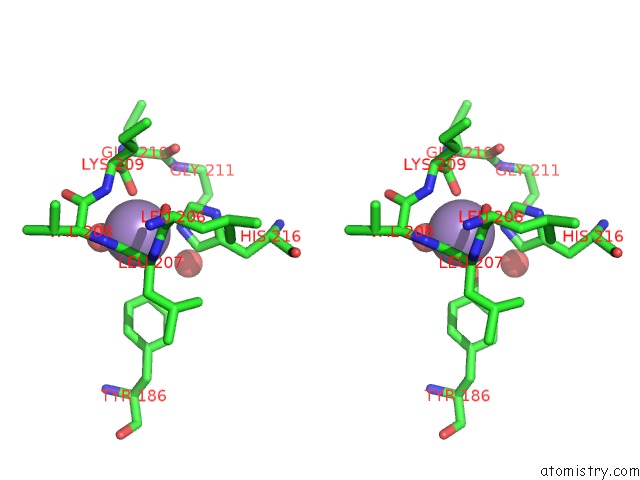
Manganese binding site 4 out of 4 in 6iht
Go back to
Manganese binding site 4 out
of 4 in the Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide
Mono view
Stereo pair view

Mono view

Stereo pair view
A full contact list of Manganese with other atoms in the Mn binding
site number 4 of Crystal Structure of Bacterial Serine Phosphatase Bound with Phosphorylated Peptide within 5.0Å range:
|
Reference:
T.Yang,
T.Liu,
J.Gan,
K.Yu,
K.Chen,
W.Xue,
L.Lan,
S.Yang,
C.G.Yang.
Structural Insight Into the Mechanism of Staphylococcus Aureus STP1 Phosphatase. Acs Infect Dis. V. 5 841 2019.
ISSN: ESSN 2373-8227
PubMed: 30868877
DOI: 10.1021/ACSINFECDIS.8B00316
Page generated: Sun Oct 6 04:53:42 2024
ISSN: ESSN 2373-8227
PubMed: 30868877
DOI: 10.1021/ACSINFECDIS.8B00316
Last articles
F in 4HUAF in 4HU9
F in 4HQJ
F in 4HT3
F in 4HLQ
F in 4HT0
F in 4HNA
F in 4HPX
F in 4HQH
F in 4HNS